The World Health Organization (WHO) declared that antimicrobial resistance is one of the top ten global public health threats facing humanity. Experts have even estimated that as many as ten million people could die each year as a result of antimicrobial resistance by 2050.
What is antimicrobial resistance, and what causes it?
Antimicrobial resistance (AMR) is when germs like bacteria, viruses, or fungi change over time and no longer respond to medicines designed to kill them. This increases the risk of disease spread, severe illness, and death. Furthermore, AMR causes antibiotics and other antimicrobial medicines to become ineffective, making infections difficult or impossible to treat.
AMR occurs naturally over time through genetic change, and antimicrobial-resistant organisms are found in people, animals, food, plants, and the environment. However, the main cause of antibiotic resistance is the misuse and overuse of antimicrobials, especially antibiotics.
When antibiotics are used, some bacteria die, but resistant bacteria survive and can multiply. The more we use antibiotics and the more people who don’t take antibiotics as directed, the higher chances that antibiotics won’t work when we need them in the future. As bacteria become increasingly resistant to antibiotics, even common infections will be difficult to treat and can become life-threatening.
More recently, the CDC’s special report on COVID-19’s impact on antimicrobial resistance in the United States concluded that the threat of antimicrobial-resistant infections grew during the pandemic. A major cause of this growth was the “…significant increase in antimicrobial use, difficulty in following infection prevention and control guidance, and a resulting increase in healthcare-associated, antimicrobial-resistant infections in U.S. hospitals.” Additionally, antibiotics were often the first option given to treat those with COVID-19, even when research showed that antibiotics were not effective.
Further deepening the problem is the lack of new antimicrobials and access to quality medicine, especially in developing countries. Other drivers include lack of clean water, sanitation, and hygiene, poor infection and disease prevention and control, and lack of awareness and knowledge.
![OECD projected global average antimicrobial resistance prevalence rates by 2030 [2]](https://www.tomorrowsworldtoday.com/wp-content/uploads/2022/09/Image3-22.jpg)
![OECD projected global average antimicrobial resistance prevalence rates by 2030 [2]](https://www.tomorrowsworldtoday.com/wp-content/uploads/2022/09/Image3-22.jpg)
Examples
Drug Resistance in Viruses
Antiviral drug resistance is an increasing concern in immunocompromised patient populations because of ongoing viral replication and prolonged drug exposure. Resistance has developed to antiviral drugs like antiretroviral drugs.
All antiretroviral drugs are at risk of becoming partly or fully inactive because of drug-resistant HIV. The increasing levels of resistance have become alarmingly high. For example, in sub-Saharan Africa, over 50 percent of the infants newly diagnosed with HIV carry a virus that is resistant to NNRTI, one class of drugs that treat HIV.
Drug Resistance in Bacteria
High rates of resistance against antibiotics frequently used to treat common bacterial infections have been observed worldwide, indicating that we are running out of effective antibiotics.
For example, Staphylococcus aureus, known as MRSA, is a type of bacteria difficult to treat because of antibiotic resistance. MRSA most often causes skin infections, but can also cause pneumonia. And, if the infection becomes severe, it can cause sepsis. According to the WHO, people with methicillin-resistant Staphylococcus aureus (MRSA) infections are 64 percent more likely to die than people with drug-sensitive infections.
Drug Resistance in Malaria
Advertisement
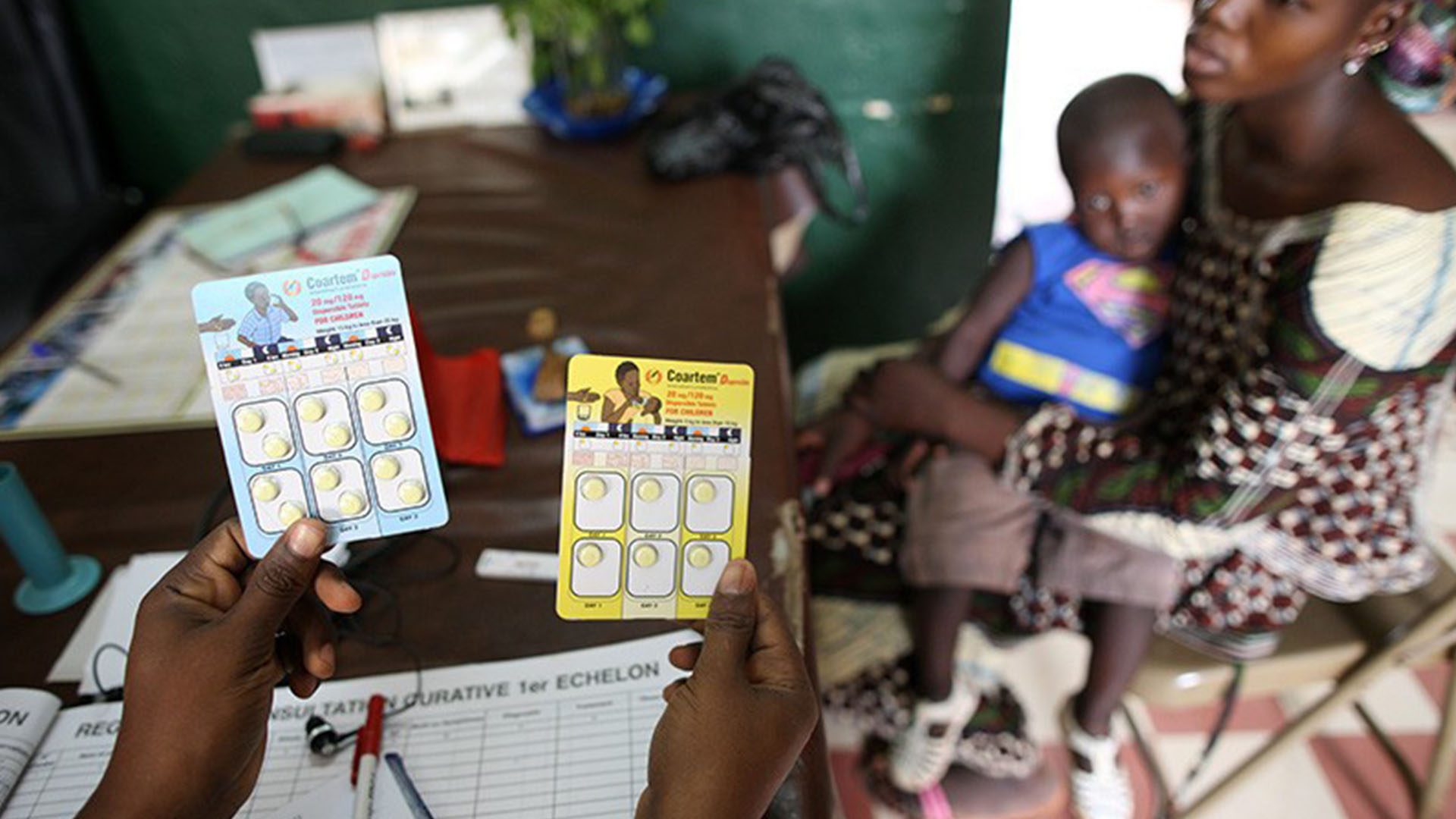

One of the greatest threats to malaria control is the emergence of drug-resistant parasites. Artemisinin-based combination therapies (ACTs) are used by most malaria-endemic countries to treat P. falciparum, the predominant species of malaria that is most likely to progress to severe forms.
Recently published evidence shows the emergence of mutations linked to partial artemisinin resistance in Rwanda. If resistance to artemisinin and ACT partner drugs continues, it could spark a major public health challenge.
How can we avoid this crisis?
AMR is a complex problem that requires a united multisectoral approach. Put simply, we can avoid this crisis by using antibiotics more carefully, developing new antibiotics and tools to treat antibiotic-resistant infections and eliminate the spread of AMR, and preventing infections. Unfortunately, these three tactics are much easier said than done. Early and appropriate treatment of infections is crucial in the fight against antimicrobial resistance. Finding a trusted general practitioner near me can ensure you get the right care at the right time.
Organizations
The WHO believes that using the One Health approach is the best solution to avoiding the spread of AMR. This approach includes partnering across sectors, population levels, and disciplines in order to achieve optimal health outcomes. Abiding by this philosophy, many organizations and partnerships have formed to address the problem of AMR.
For example, in 2015, countries committed to the Global Action Plan on Antimicrobial Resistance. To guarantee global progress, the countries in the partnership must ensure the cost and implementation of national action plans across sectors to ensure sustainable progress.
Similarly, the United Nations (UN) created the Interagency Coordination Group on AMR in 2018. In 2019, the group submitted its report “No time to wait: Securing the future from drug-resistant infections” to the UN Secretary-General. The recommendations proposed in this document are now being implemented, such as providing equitable and affordable access to quality antimicrobial agents and investing in new antimicrobial medicines, diagnostics, vaccines, and waste management tools.
Scientists & Companies
Many scientists and companies are testing alternative ways to prevent AMR. For example, researchers have found that a naturally occurring compound has the potential to kill drug-resistant bacteria. The compound, known as hydroquinine, is found in the bark of some trees. It is already known to be an effective agent against malaria, but new findings suggest that hydroquinine could be used in new antimicrobial drugs to combat infections.
As for companies, Janssen, a pharmaceutical company owned by Johnson & Johnson, is leveraging one of nature’s ways to control bacteria: bacteriophages. Bacteriophages, also known as phages, are any group of viruses that infect and kill bacteria. Bacteriophages shred the bacteria’s DNA so that it can’t survive.
As Dr. James Mersobn, Ph.D., Janssen’s Global Therapeutic Area Head of Infectious Diseases, told us at Tomorrow’s World Today, “We believe this is going to be a fundamental new way of how we can manage antimicrobial resistance.”
For more information about antimicrobial resistance and the future of medicine, we visited Atara’s and Janssen’s laboratories. Check out what we discovered on Tomorrow’s World Today below.



